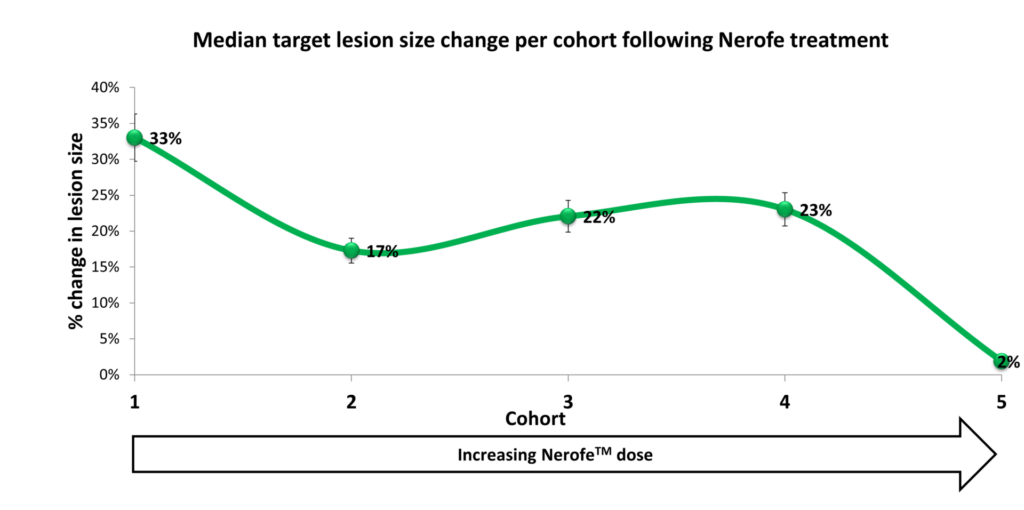
I. Anti-proliferative
In 3 patients with elevated serum EGF levels, levels decreased to normal values (Remaining patients entered trial with normal EGF levels)

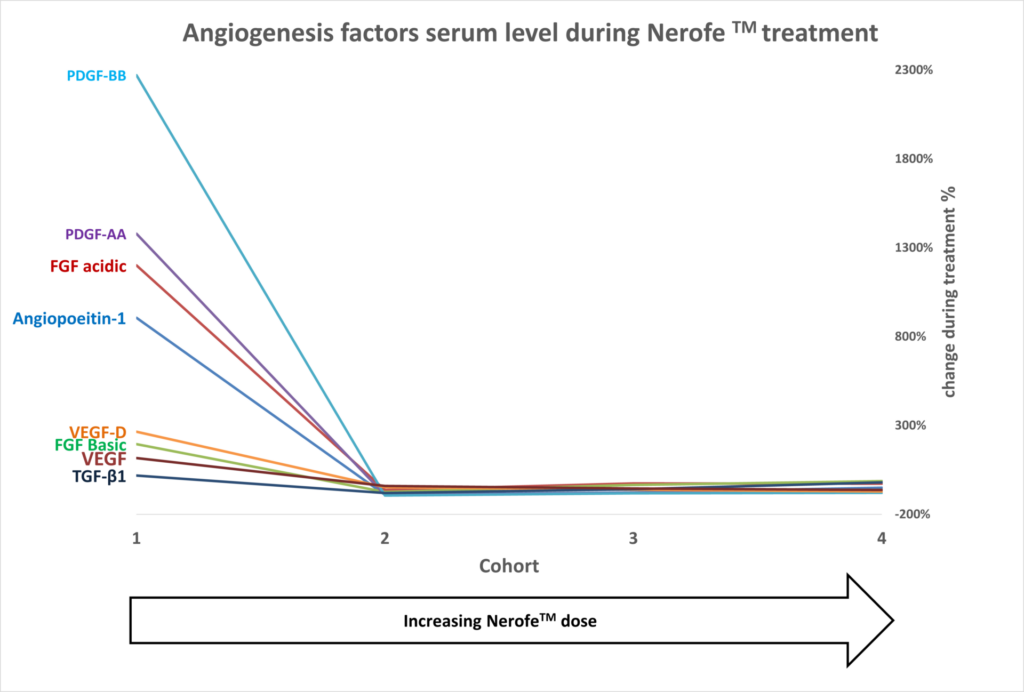
II. Anti-angiogenic
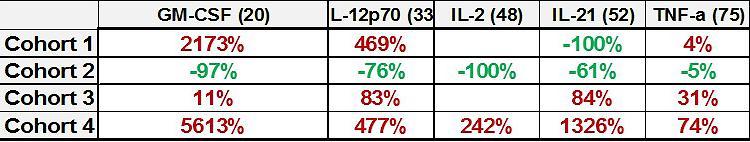
III. Induction of the Immune System
Selective biomarker for efficacy
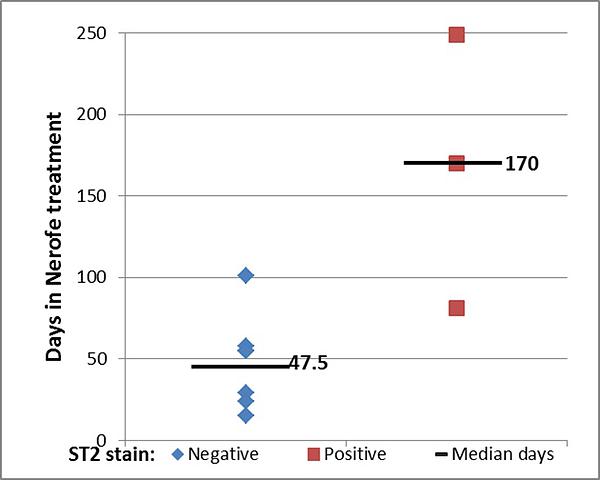


In 3 patients with elevated serum EGF levels, levels decreased to normal values (Remaining patients entered trial with normal EGF levels)



